Difference between revisions of "Pressure-Temperature"
m (→Decarboxylation) |
m (→Methods) |
||
| Line 13: | Line 13: | ||
<hr> | <hr> | ||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
= Temperatures = | = Temperatures = | ||
Revision as of 06:47, 14 June 2023
YouTube ... Quora ...Google search ...Google News ...Bing News
Contents
Temperatures
The temperature at which a compound vaporizes is called its boiling point. The boiling points of different compounds vary, so by controlling the temperature, the desired compounds can be separated from the plant material.
For example, to distill THC, the temperature would be set to 157°C (315°F). This would cause the THC to vaporize and rise up the distillation column, where it would be condensed back into a liquid. The other cannabinoids and terpenes would remain in the plant material.
The temperature at which a compound is distilled can also affect its purity. If the temperature is too high, the compound can be degraded. If the temperature is too low, the compound may not be completely vaporized. It is important to use a precise temperature control system when distilling cannabis. This will help to ensure that the desired compounds are separated from the plant material and that they are distilled at the correct temperature to maintain their purity.
- Cannabinoids:
- THC: 157°C (315°F)
- CBD: 180°C (356°F)
- CBG: 126°C (259°F)
- Terpenes:
- Limonene: 177°C (350°F)
- Linalool: 198°C (388°F)
- Pinene: 156°C (312°F)
- Solvents:
- Ethanol: 78.5°C (173.3°F)
- Butane: −0.5°C (31.1°F)
Short Path Distillation
Short Path Distillation: is a technique used to separate and purify different compounds in cannabis oil, such as cannabinoids and terpenes. The process involves heating the cannabis extract under vacuum conditions to reduce the boiling points of the desired compounds. The following are approximate temperature ranges used in Short Path Distillation:
- Heads Fraction: The heads fraction typically contains volatile compounds with lower boiling points, such as terpenes. The temperature range for collecting the heads fraction is around 248 to 302 degrees Fahrenheit (120 to 150 degrees Celsius).
- Body Fraction: The body fraction consists of the desired cannabinoids, including THC and CBD. The temperature range for collecting the body fraction is approximately 302 to 392 degrees Fahrenheit (150 to 200 degrees Celsius).
- Tail Fraction: The tail fraction usually contains heavier compounds and impurities. The temperature range for collecting the tail fraction is around 392 to 572 degrees Fahrenheit (200 to 300 degrees Celsius).
Crystallization
In certain cases, a final step is taken to separate THC from CBD. Crystallization is a common method. A reactor vessel is filled with feedstock and a solvent which is chilled slowly from 60 degrees Celsius to minus-20 degrees Celsius. A slurry results and that is transferred to a Nutsche filter dryer to produce pure, dried crystals. The Nutsche filter is a jacketed vessel in which the temperature is controlled with a circulating hot oil unit. The process results in a 98% or higher purity of the CBD or THC product.
Pressure-Temperature
- Boiling Point of THC - Bleaker ... nomagraph and spreadsheet below
- Raoult's Law and Ideal Mixtures of Liquids | Chemistry LibreTexts
- Vapour Pressure-Composition Diagram for Ideal Solutions | Minia
- 8.9: Distillation - Chemistry LibreTexts
- 4.5: Separating Volatile Solutions - Chemistry LibreTexts
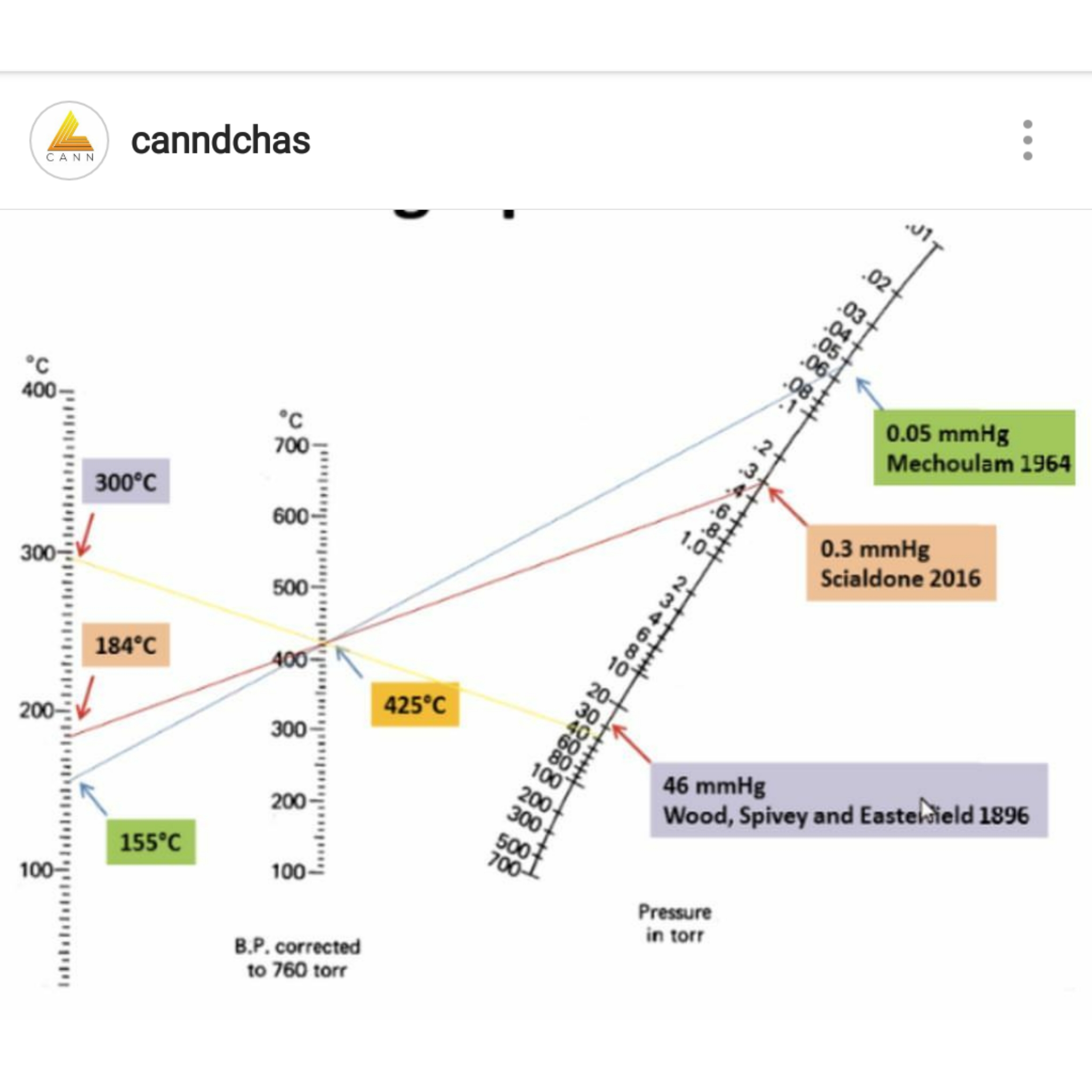
Nomograph: You find those numbers on the lines and draw a straight line between them. Where that line crosses the other line is the answer to your problem.
The chart (and the math that created it) was the basis for the computation for the pressure curve really I created. If you observe two known points of a boil at two different pressures then the math that created that curve chart you posted can be used to extrapolate all the rest of the points along the curve. The chart you posted uses mmHg for the pressure unit; .01 mmHg listed as the top pressure value = ten microns. I use microns as a unit of reference. The math gives crude estimates.
If you take the two known points I used you can plot it on that chart. “Boiling” is somewhat a subjective term at these pressures. I used the bottom of the bottom of the cold finger glass of the sublimation apparatus to judge when accumulation became noticeable as the “boiling” point. The two data points used in the calculation for the D9 THC chart were 120C at ¾ of one micron, and 200C at 20 microns. You can plug these points into the chart above and see how high the boil would be at room pressure which is 760,000 microns (the middle of your chart is corrected to 760 Torr = 760,000 microns). A theoretical perfect vacuum is zero microns.
From that chart you posted it is also possible mathematically to compute the enthralpy of evaporation. This number means very little to the daily refinement but it does offer clues about what is going on inside the rig when it runs and it helped me to figure out my horizontal distillation set up. I run the distillation pass horizontally via kugelrohr bulbs but use a heating mantle and stir bar instead of a formal oven. The temps run much lower to pull across distillate this way and the high numbers suggested this would work better than a traditional vertical distillation. It does. It is because you are not sinking all that heat into the uptake path in a horizontal mode.
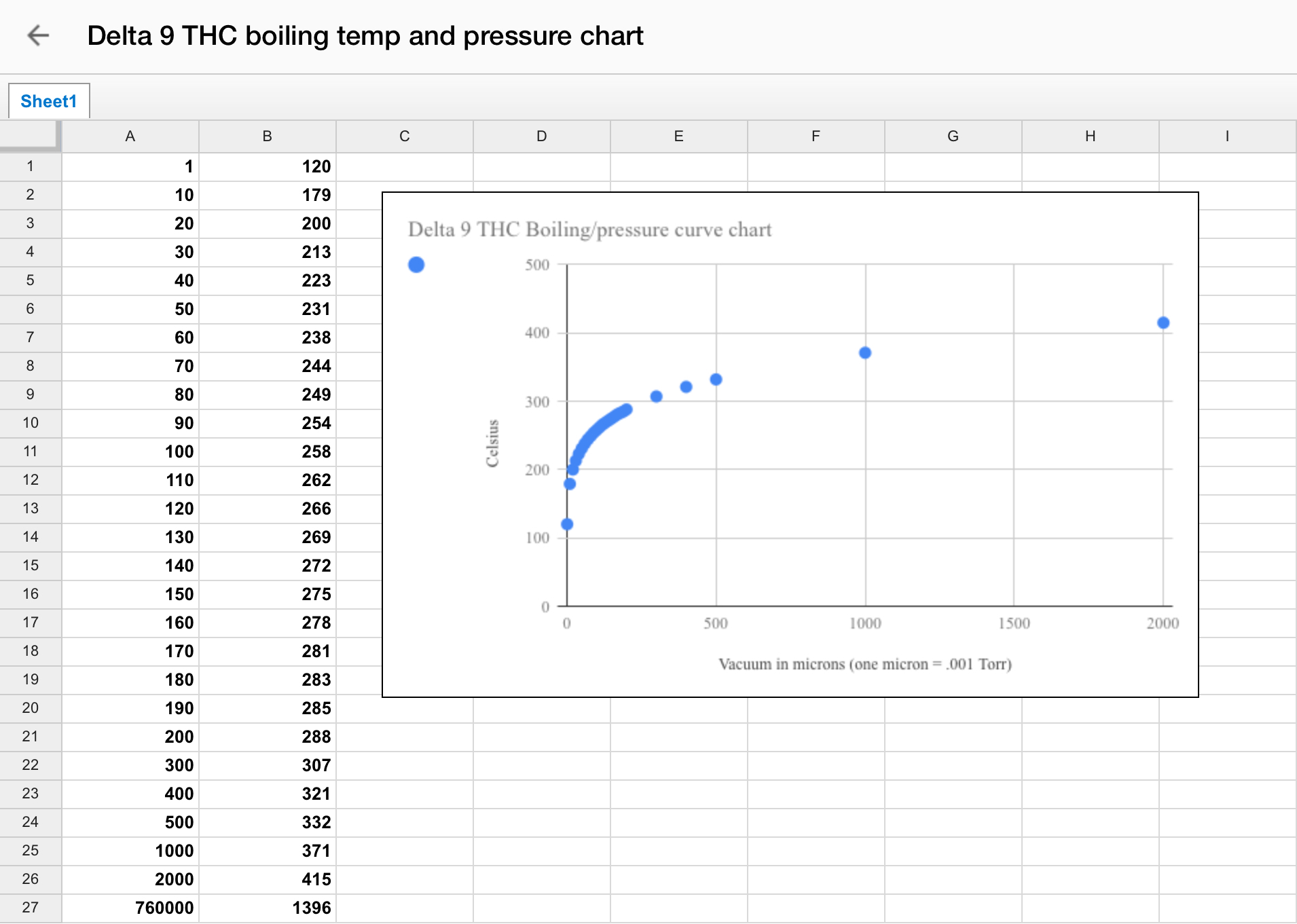
Pressure-Temperature Nomograph
To fully understand distillation, one must consider the vapor pressure vs. composition plots for a hypothetical mixture at some arbitrary temperature at which both liquid and gas phases can exist, depending on the total pressure. You can use tools such as the Pressure-Temperature Nomograph Interactive Tool to calculate a boiling point or pressure using the Antoine Equation. You will need to adjust the temperature and vacuum settings according to the boiling points of different cannabinoids. For example, THC boils at around 157°C (315°F), while CBD boils at around 180°C (356°F).
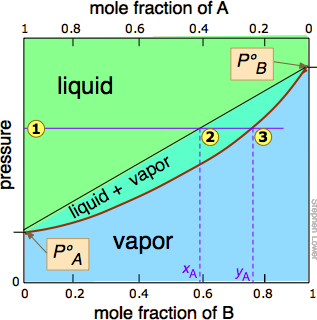
Vapor pressure vs. composition plots are used to understand the behavior of mixtures of two volatile liquids. These plots show the relationship between the vapor pressure and the composition of a mixture at a given temperature. The vapor pressure of a mixture is determined by the vapor pressures of its individual components and their mole fractions in the mixture. Raoult's Law states that the partial vapor pressure of each component in an ideal mixture is equal to the product of its mole fraction and its vapor pressure as a pure substance.
In an ideal mixture, the intermolecular forces between two molecules of one component must be exactly the same as the intermolecular forces between a molecule of one component and a molecule of the other component. This is why mixtures like hexane and heptane get close to ideal behavior. They are similarly sized molecules and so have similarly sized van der Waals attractions between them.
A vapor pressure vs. composition plot can be constructed by plotting the total vapor pressure of the mixture against the mole fraction of one of its components. The plot will show two curves: one for the liquid phase and one for the vapor phase. The liquid composition curve lies above the vapor composition curve. The intersection of a horizontal line representing the total pressure with these two curves defines the compositions of the liquid and vapor phases in equilibrium at that pressure.
Vapor pressure composition plot vs Pressure-temperature chart
A vapor pressure composition plot is a graph that shows the relationship between the vapor pressure and the composition of a mixture at a given temperature¹. It is used to understand the behavior of mixtures of two volatile liquids. The plot will show two curves: one for the liquid phase and one for the vapor phase. The liquid composition curve lies above the vapor composition curve⁵.
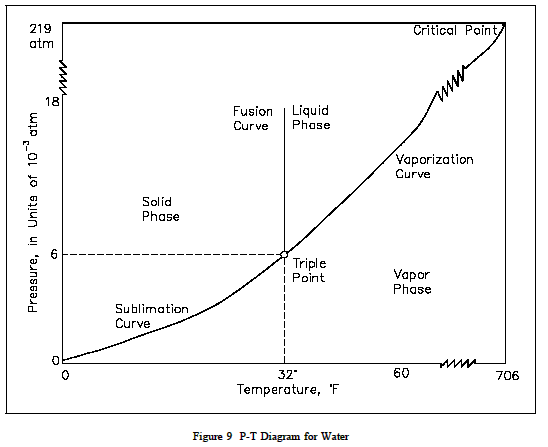
On the other hand, a pressure-temperature chart is a graph that shows the relationship between the pressure and temperature of a substance. It is used to understand how the pressure of a substance changes with temperature. For example, a pressure-temperature chart for water would show how the vapor pressure of water changes with temperature².
The main difference between these two types of graphs is that a vapor pressure composition plot shows how the vapor pressure of a mixture changes with its composition, while a pressure-temperature chart shows how the pressure of a substance changes with temperature.
Molecular Weight - mole
- Molecular Weight: Molecular weight is the mass of a molecule that is calculated by adding the atomic weights of its constituent atoms. It can be expressed in terms of atomic mass units (amu) or Daltons (Da). Molecular weight is used in chemistry to determine stoichiometry in chemical reactions and equations.
- Mole: is a unit of measurement used in chemistry to measure the amount of a substance in the International System of Units (SI). The quantity amount of substance is a measure of how many elementary entities of a given substance are in an object or sample. It is similar to other units of measurement like a dozen or a pair, but instead of representing 12 or 2 items, one mole represents 6.02214076 × 10^23 particles. This number is known as Avogadro's number and is used to convert between the number of atoms or molecules and the mass in grams. The mole is used as a unit of measurement because it provides a consistent way to convert between the number of atoms or molecules and the mass in grams. This makes it easier to perform calculations and understand the amounts of substances involved in chemical reactions.
In a vapor pressure vs. composition plot, the mole is used to represent the number of particles in a mixture. The plot above shows the relationship between the vapor pressure and the composition of a mixture at a given temperature. The vapor pressure of a mixture is determined by the vapor pressures of its individual components and their mole fractions in the mixture. A mole fraction is the ratio of the number of moles of one component to the total number of moles in the mixture.
Tips
Here are some additional tips for distilling cannabis:
- Use fresh, high-quality cannabis material.
- Use a clean, well-maintained distillation apparatus.
- Follow the manufacturer's instructions for your distillation apparatus.
- Monitor the temperature carefully during distillation.
- Collect the distillate in a clean, dry container.